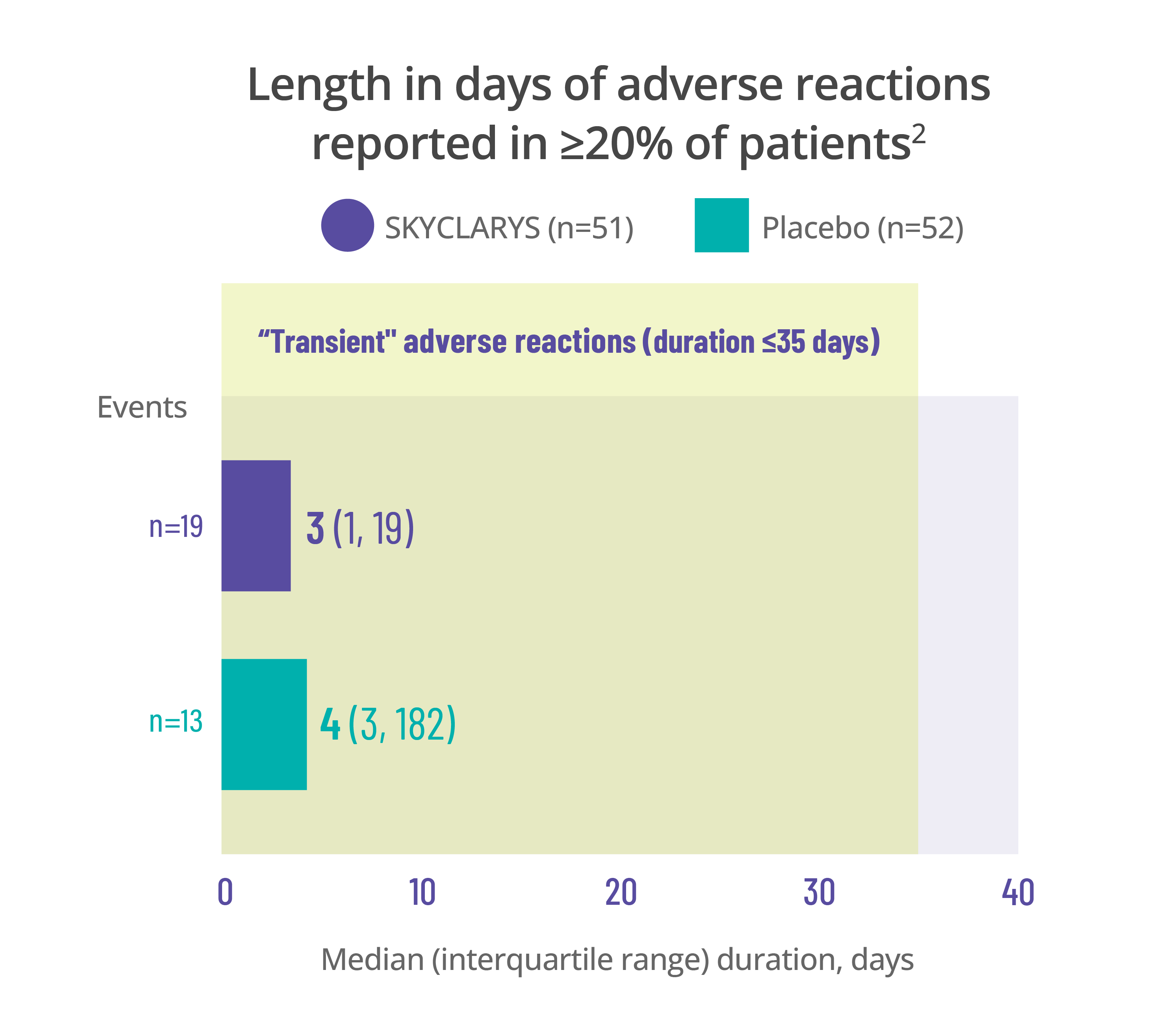
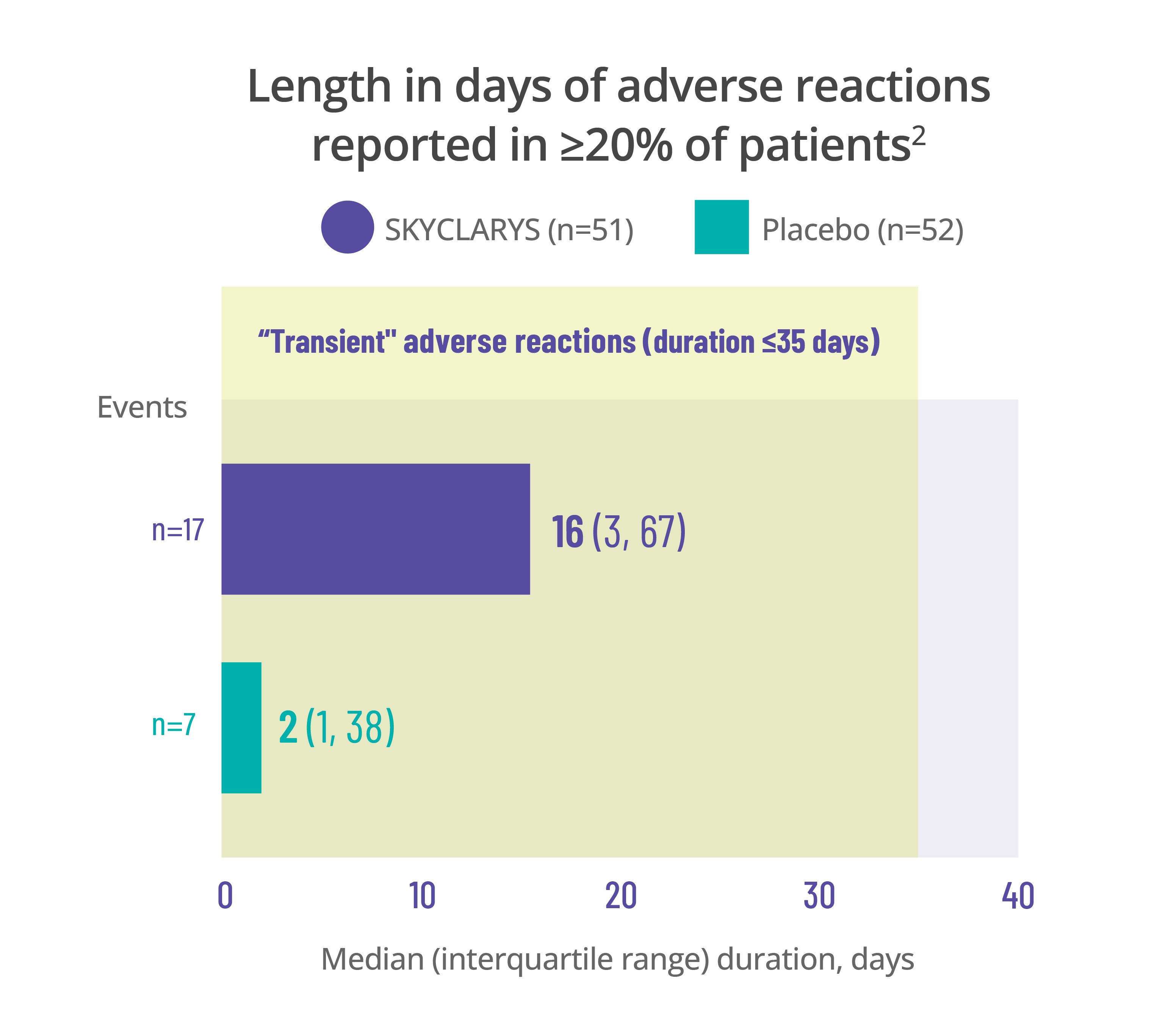
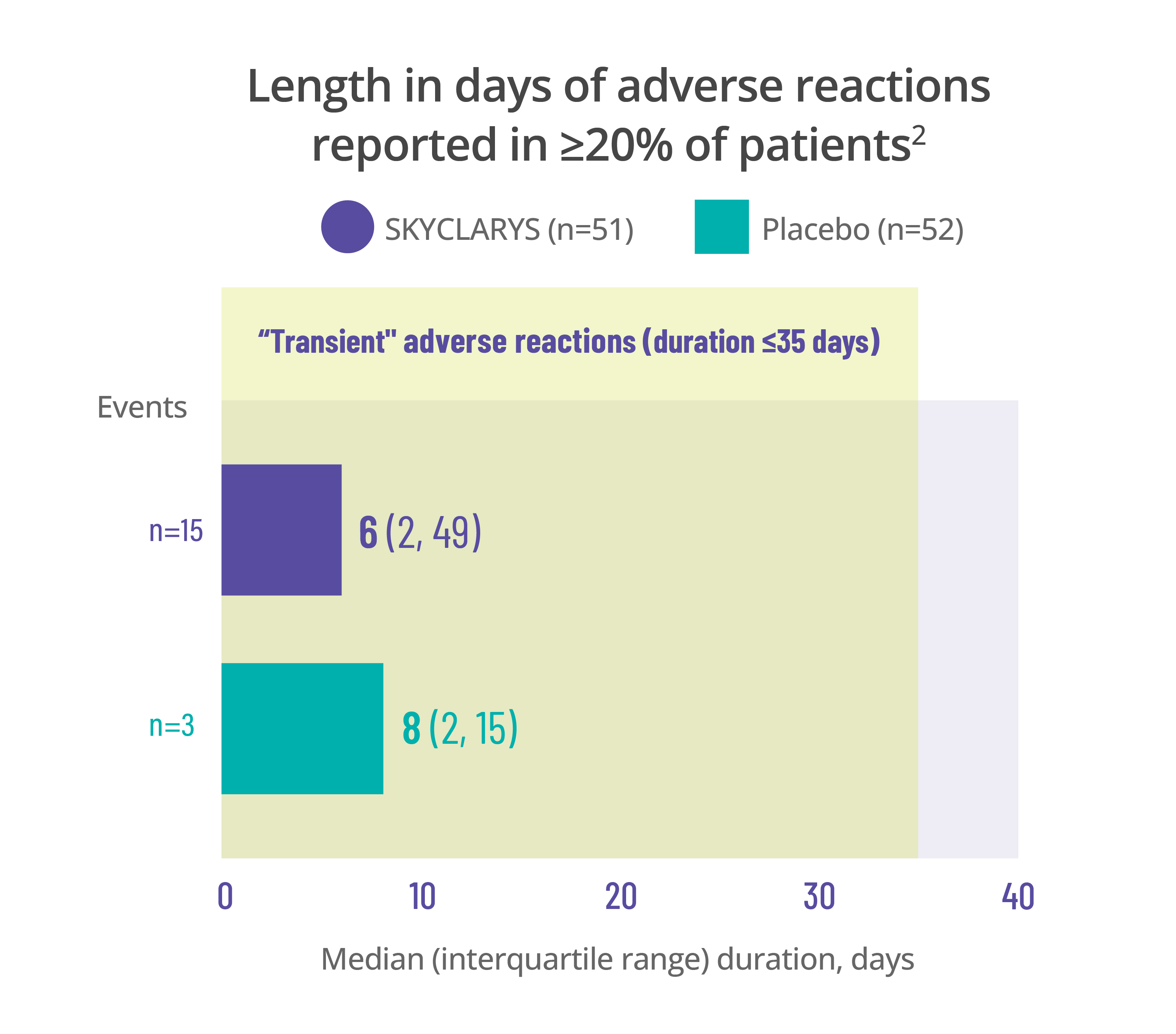
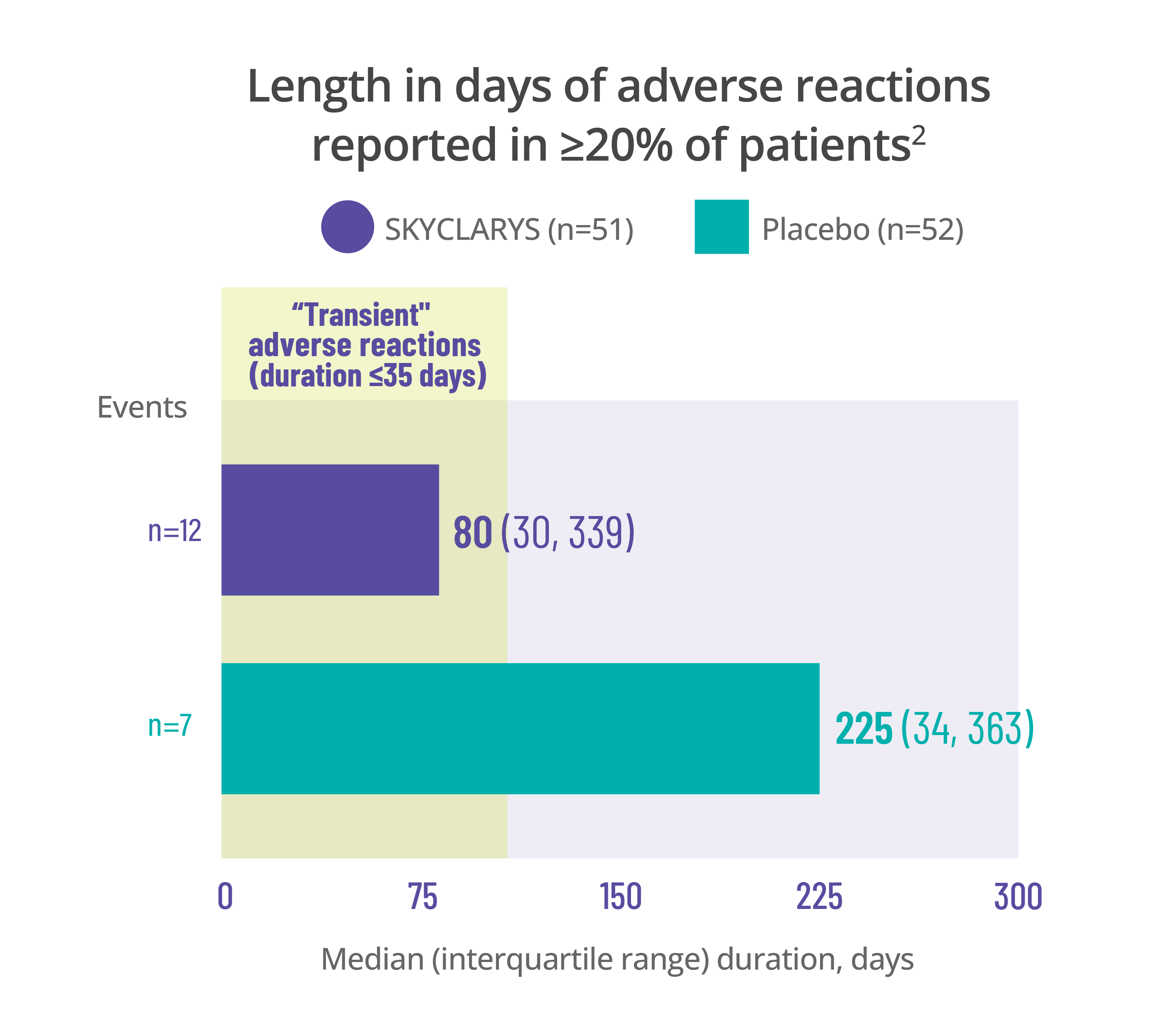
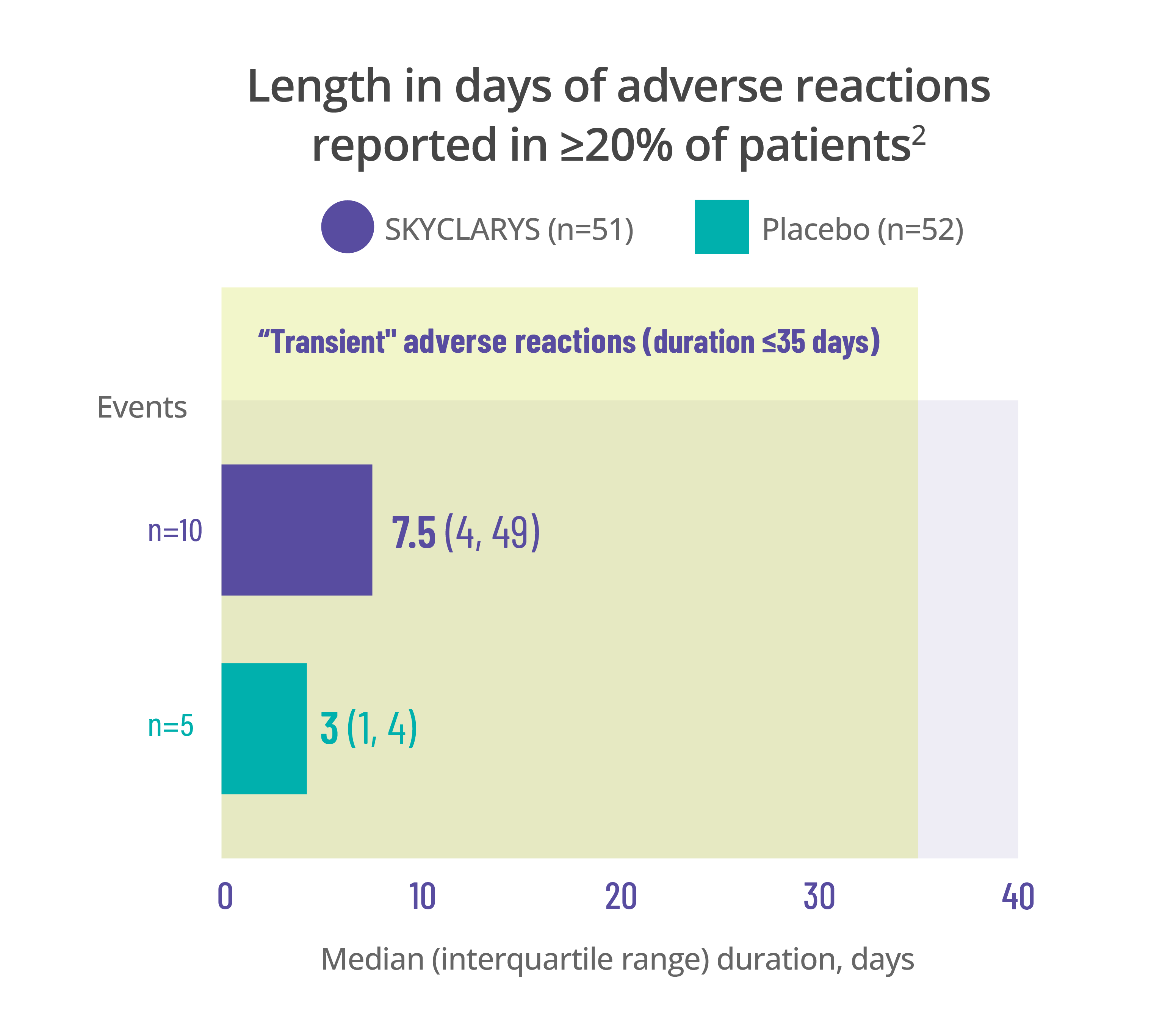
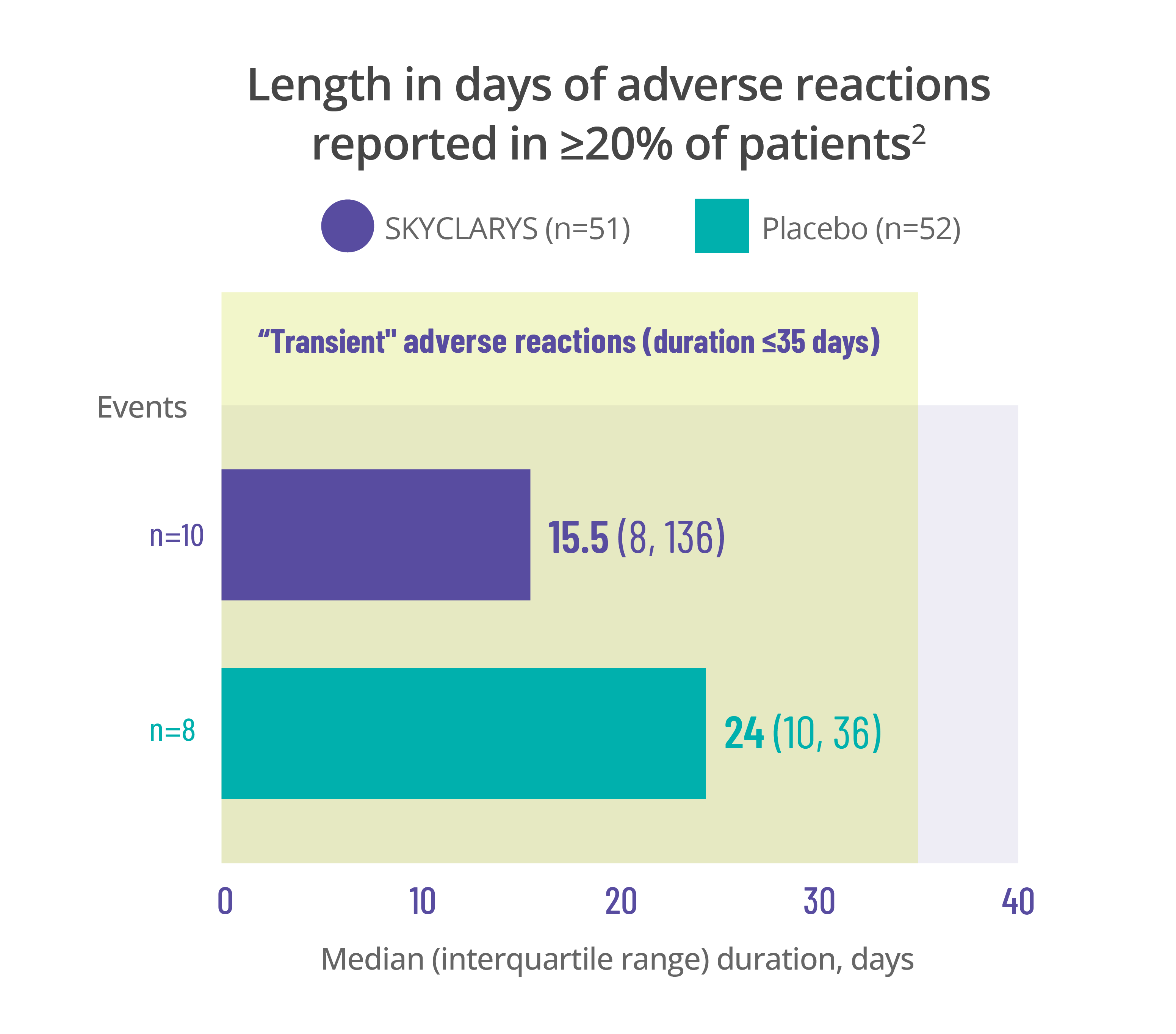
Most common adverse events
were transient, resolving in ≤35 days1
Data for the duration of adverse reactions is represented by median and quartile values:
- The median value divides the data set in half, meaning 50% of patients saw their adverse reaction resolve before that point and 50% after
- The first quartile is the point before which 25% of patients experienced resolution
- The third quartile is the point before which 75% of patients experienced resolution
Dividing data into quartiles helps illustrate the distribution of data points and variability of the duration of adverse reactions.
Data here is limited to the duration of the MOXIe trial. Remember that adverse reactions can occur at any time, even after 1 year.
It is important to discuss adverse reaction duration, including potential liver enzyme elevations, with your patients to help appropriately set treatment expectations1,3
Length in days of adverse reactions reported in ≥20% of patients2
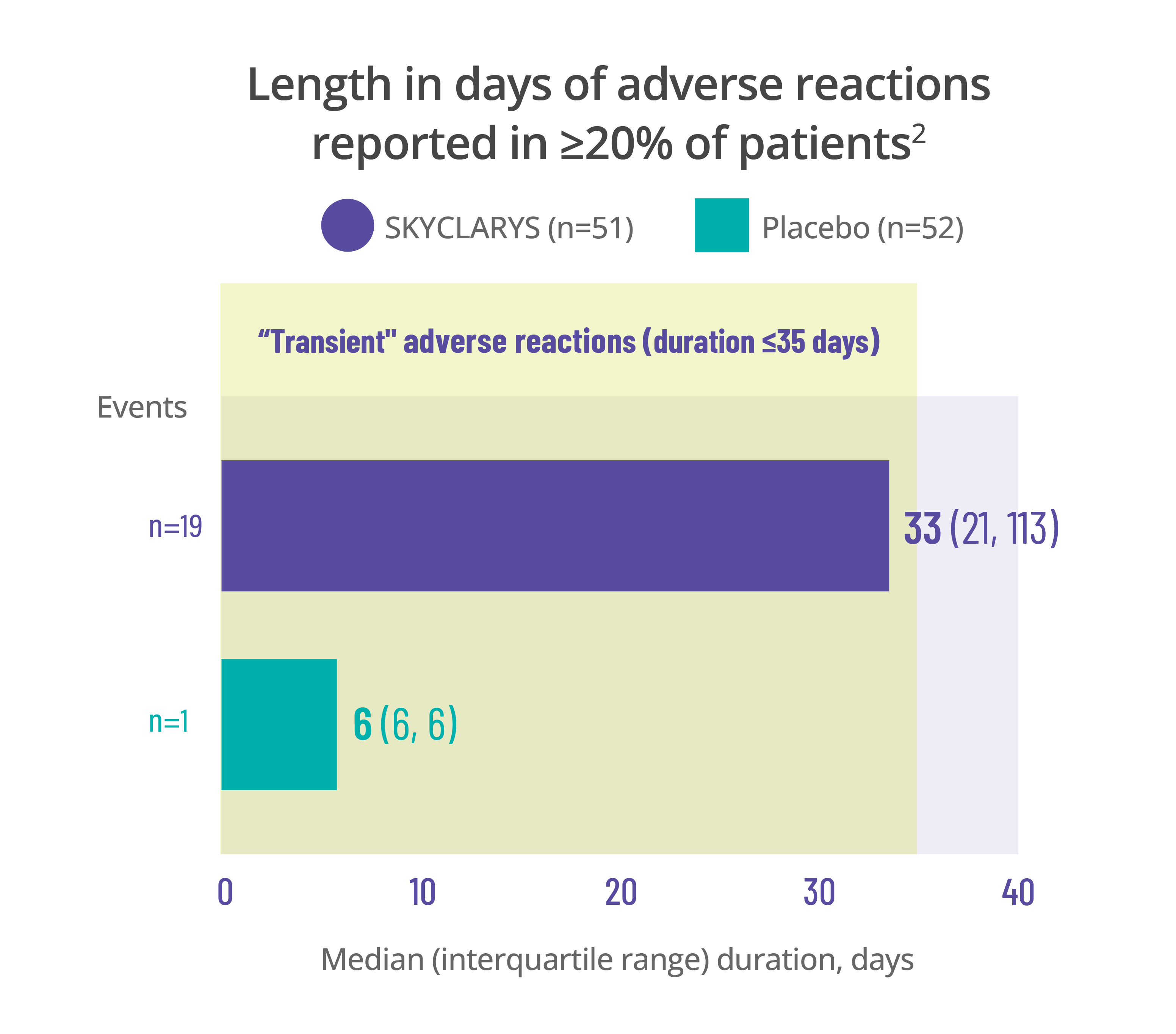
Elevations in ALT and AST observed in the MOXIe trial were generally transient and there were no reported cases of liver injury1,3,4